There is a growing emphasis on functional outcomes and quality of life in sarcoma patients. Proton therapy offers an innovative approach to achieve these goals.
Following diagnosis, local treatment is approached through wide excision of the tumor with or without radiotherapy to eradicate adjacent tumor cells. For all sarcoma patients, local tumor control ranges from 30% to 100%, but that data represents a broad spectrum of sarcoma subtypes and treatments. Certain tumors that are high grade, large, and/or located deep under the skin are associated with the highest risk for recurrence. Also, compared to younger individuals (i.e., those <55-60 years old), tumors in older patients tend to relapse more often.
At the time of the cancer surgery, obtaining wide excision margins around the tumor may be difficult as the cancer infiltrates tissue near critical organs. If excision is attempted and there is a high chance of residual tumor, the surgeon will often refer a patient for radiation therapy to treat the remaining microscopic cancer cells. An alternative approach is to give radiation before surgery (neoadjuvant or preoperative radiation), shrinking the tumor and making complete excision more likely. A less ideal situation is when a large or aggressive tumor simply cannot be removed without an amputation or radical surgery. In the absence of surgery, these patients require a very high dose of radiation for a chance of cure.
Combined modality treatment (meaning surgery and radiation) is very valuable in controlling sarcomas and clinical trials have established the benefits of radiotherapy including limb preservation, the avoidance of disfiguring surgery, and increased local tumor control preventing a second surgery and improving overall survival. However, combined modality therapy involves significant risks, with complications affecting up to a third of patients. These include problems with wound healing, fibrosis of the skin, muscle weakness, joint stiffness, and edema (swelling). These wound complications may necessitate reoperation or amputation.
There is a growing emphasis on functional outcomes and quality of life in sarcoma patients. Proton therapy offers an innovative new approach to achieve these goals. As of 2020 there are 80 proton therapy centers in the world, including 35 centers in the United States – the University of Florida Health Proton Therapy Institute among them – fitted with the gantry systems necessary to address the diverse anatomical presentation of sarcoma. Due to unique physical characteristics of protons which cause them to stop within tissue, proton therapy can provide highly conformal radiation with minimal or no dose to normal organs. For instance, when a sarcoma is located in the abdomen or retroperitoneum, proton therapy may be utilized to save kidneys from the damaging effects of radiation. If the intestines can be avoided, there is less nausea during treatment.
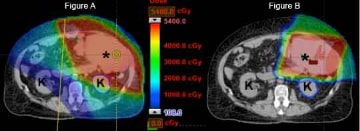
In sarcomas arising in the arm and leg, proton therapy may be utilized to completely spare the tissue flap a surgeon uses to repair the skin following tumor excision, thus promoting a normal healing environment. Also, when a sarcoma abuts a bone, patients have a well-documented risk of fracture related to surgery and radiation. By decreasing the radiation dose to bone, conformal proton therapy may reduce the incidence of a broken bone in the future. By avoiding any exit dose, other theoretical advantages of proton therapy in lower extremity sarcoma treatment include avoiding radiation dose to the groin and perianal region. These areas are exquisitely radiosensitive and skin irritation during treatment may force undesirable delays in treatment. By avoiding the testes and ovaries in young patients, radiation oncologists can use proton therapy in an attempt to maintain patient fertility. Finally, by decreasing the normal tissue dose compared to photon therapy, proton therapy should minimize the risk of radiation-induced secondary cancers in survivors. Based on these issues, proton therapy has been successfully utilized in conjunction with surgery to treat sarcoma patients at UF Health Proton Therapy Institute and other centers around the world. As overall survival rates increase and long-term follow-up data builds, the benefit of proton therapy in terms of late effects may become even more prominent in our patients.
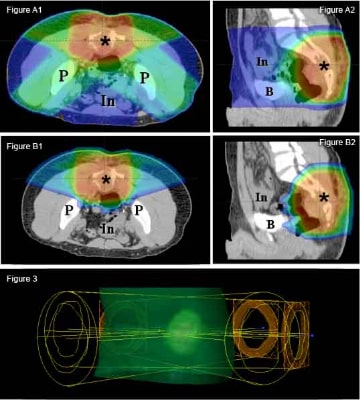
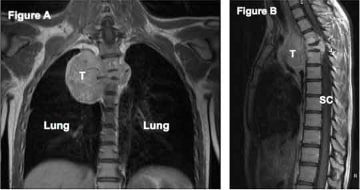
The advantages of using proton therapy to spare normal tissue extend to tumors unsuitable for surgery. In sarcomas arising from or near the spine, complete resection is often a challenge. Delivery of an adequate radiation dose is limited by surrounding radiation-sensitive normal tissues, including the spinal cord, intestines, lungs, heart, and kidneys. For example, in a tumor abutting the spinal cord, the clinician is often forced to balance inadequate local control with the unacceptable risk of serious side effects. The precise dose delivery of proton therapy is ideal for this situation.
Many high risk soft-tissue sarcomas and bone sarcomas are treated with chemotherapy that may interact unfavorably with radiation. For instance, studies have described more skin irritation and bone fractures following combined radiation and chemotherapy treatment. By minimizing the overlap of chemotherapy and radiation in normal tissue, proton therapy has an exciting potential role in synergistic treatment regimens.
Summary
The University of Florida has been a leader in research and treatment of sarcoma for over 40 years. This continues today at the University of Florida Health Proton Therapy Institute where sarcoma in adults and children is a top priority. Our physicians are always glad to evaluate any patient who may need radiation therapy as part of the treatment plan. We work closely with an experienced team of sarcoma surgeons and medical oncologists.
We strongly recommend your evaluation for proton therapy takes place before any surgery or chemotherapy: A multidisciplinary plan should be designed in advance as treatment decisions may impact your eligibility for proton radiation.
Please feel free to contact us via the web, e-mail, or phone if you have any questions. Sometimes we can tell you whether or not you might benefit from proton therapy from this first inquiry, but often we will need further information before making a final recommendation and we will work with you to gather that information.



